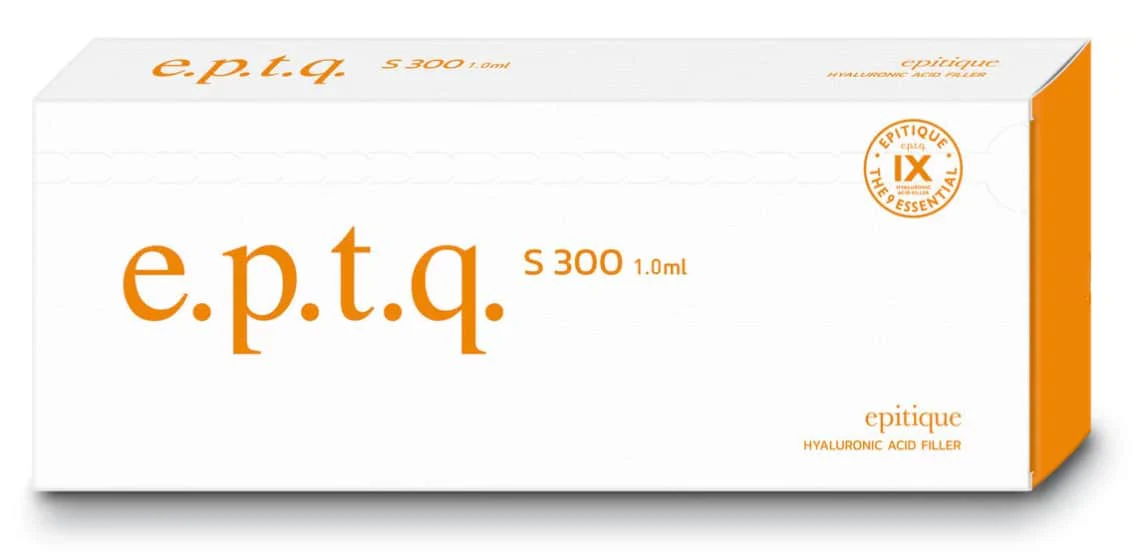
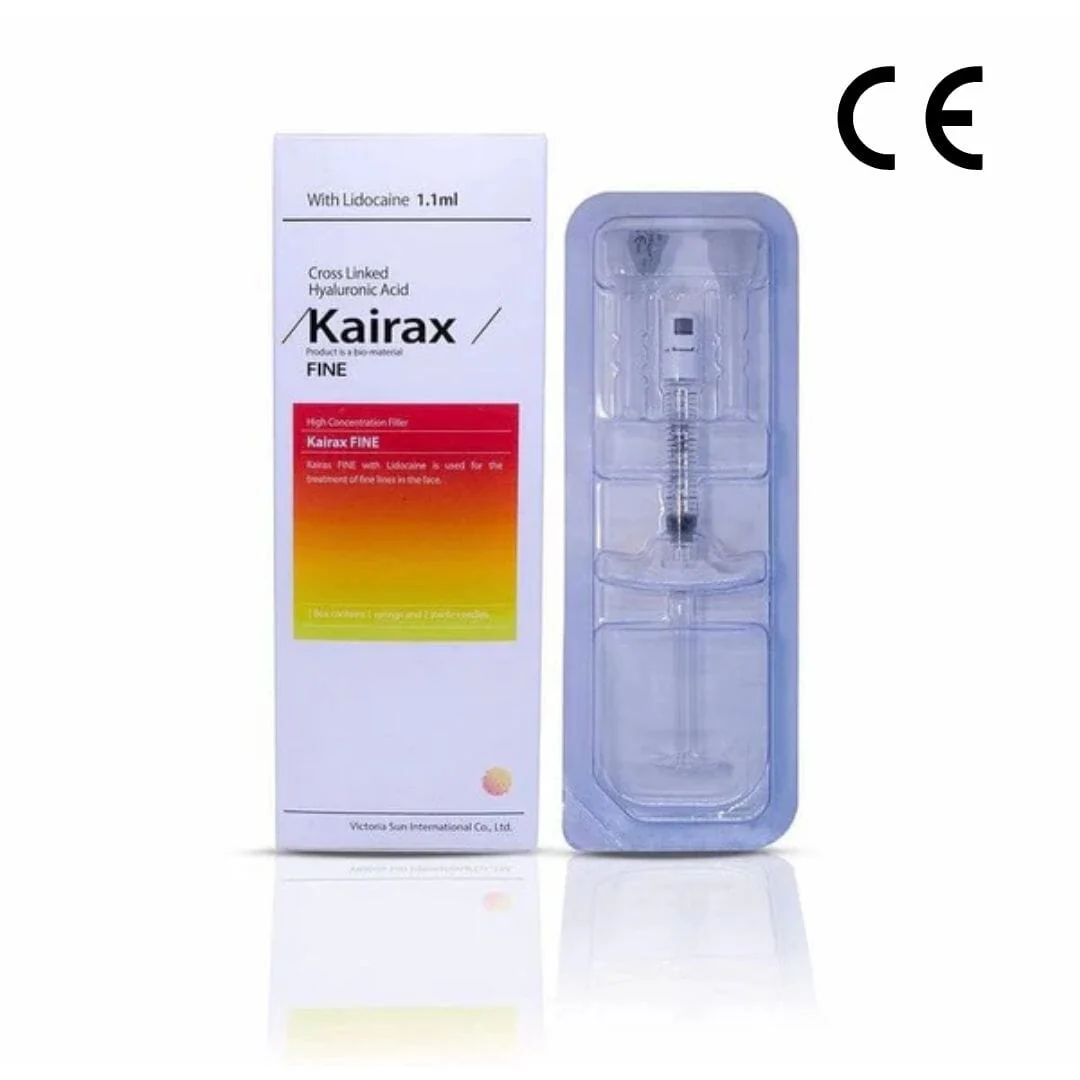
e.p.t.q.® Lidocaine S 300 is a CE-certified, mid-density hyaluronic acid (HA) filler formulated for mid-to-deep dermal implantation. Each 1.1 mL syringe contains 24 mg/mL of cross-linked, non-animal HA and 0.3% lidocaine for a comfortable injection experience. Manufactured in South Korea via a rigorous “9 Process” protocol, it delivers natural volume restoration with an impressive initial effect.
Key Attributes
- High-Purity HA: Optimized for minimal edema and extended duration
- Superior Viscoelasticity: 300 Pa·s viscosity index for stable molding and lift
- Homogeneous Particle Technology: Ensures uniform gel structure and cohesiveness
- BDDE-Free Crosslinking: Negligible residual catalyst; endotoxin < 0.1 EU/mL
- Patient Comfort: pH- and osmolarity-matched to skin; integrated lidocaine minimizes pain
Indications
- Correction of moderate-to-deep wrinkles and folds (e.g., nasolabial, marionette)
- Facial augmentation and contouring (cheeks, chin, jawline)
- Lip enhancement
Administration
- Needles: Two 27 G × ½″ (13 mm) ultra-thin wall needles included
- Injection Depth: Mid to deep dermis
- Technique: Linear threading or bolus deposition by a trained professional
- Volume per Session: Tailored to treatment area and patient anatomy
Efficacy & Duration
- Onset: Immediate volumizing effect
- Duration: 6–8 months of sustained correction
Safety & Tolerability
- Low incidence of immune reactions or delayed hypersensitivity
- Transient, mild side effects: redness, swelling, tenderness at injection sites
- Single-use only; do not re-sterilize or combine with other products
Storage & Handling
- Store at 2–25 °C; protect from direct sunlight, freezing and heat
- Shelf life: 24 months from manufacture
Specifications
- HA Concentration: 24 mg/mL
- Lidocaine: 0.3%
- Viscosity Index: 300 Pa·s
- Syringe Volume: 1.1 mL (Lidocaine S 300); 1.0 mL (non-lidocaine version)
- Manufactured: South Korea
e.p.t.q.® Lidocaine S 300 combines advanced HA cross-linking with patient-friendly lidocaine to deliver reliable, natural-looking volumization and wrinkle correction.
Disclaimer:
Bruno Dermal Filler products are strictly intended for use by licensed medical and aesthetic professionals. Not suitable for self-administration or use by untrained individuals.